Project manager
Details
EU Horizon 2022 Disease
Duration01.05.2023 - 30.04.2027

MITI2 - Gut Microbiota-Induced Tregs for Inflammatory-Bowel-Disease (IBD) Immunotherapy
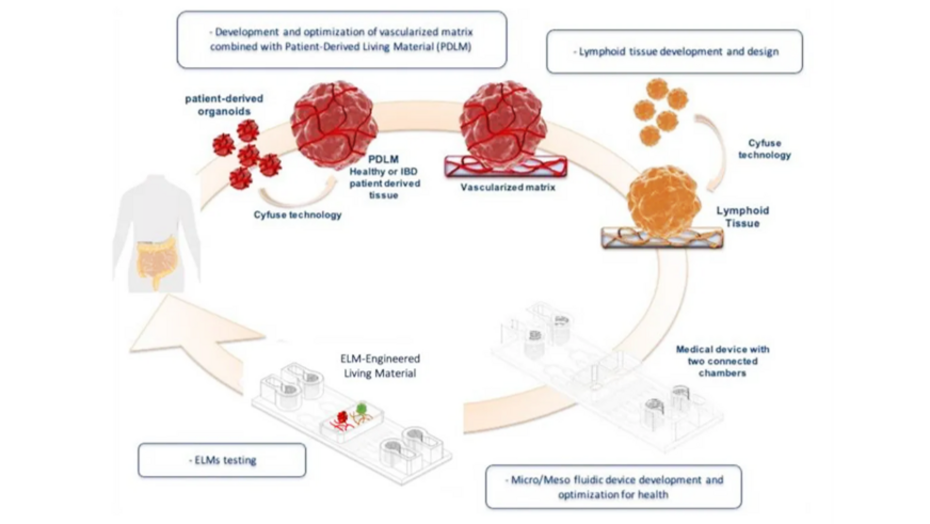
The project focuses on the regulatory properties of a subset of microbiota-specific TR1-like regulatory T (Treg) cells, for which we have already shown an unprecedented association with the clinical outcome of patients in various inflammatory diseases, for a therapeutic use in inflammatory bowel diseases (IBD). IBD is a disabling chronic inflammatory process that affects young individuals and causes many life-altering symptoms, and represents a risk factor for colon cancer. Existing treatments are complex, with most people requiring lifelong medications as well as dietary and lifestyle modifications, and some requiring surgery. In this context, the development of new therapeutic approaches appears essential and immunotherapy and cell-based therapy are particularly promising strategies for this disease. Teams from Nantes have a strong expertise in the field of human immunology, mucosal immunology and immunotherapeutic strategies applied to various pathological conditions, including gut inflammatory diseases. They recently identified a novel microbiota-induced Treg subset, associated with good prognosis in IBD patients, thus representing a promising candidate for innovative immunotherapeutic approaches. Based on the limitation to develop immunotherapy approach for human diseases by using animal models due to immune system specificities/differences and ethical considerations, we opted for the development of an ex vivo human preclinical model that will reconstitute the physiological complexity of the human gut. Teams from Strasbourg have a strong experience and already set-up models of organoids in different pathological systems, that will perfectly fit to be used as ex vivo preclinical models. This project aims thus at providing a pre-clinical package including i) the proof of concept that a cellular immunotherapy using the identified Tregs subset represents a treatment for IBDs and ii) the reglementary pre-clinical in vitro and in vivo toxicity.